 Science Daily has noted IVC filter use has decreased following an FDA advisory. IVC filter use began declining last year after the National Institutes of Health noted in June 2016 that IVC filters carry real risks. IVC filter injury lawsuits may also be slowing down the sale and use of these blood clot filters.
Science Daily has noted IVC filter use has decreased following an FDA advisory. IVC filter use began declining last year after the National Institutes of Health noted in June 2016 that IVC filters carry real risks. IVC filter injury lawsuits may also be slowing down the sale and use of these blood clot filters.The NIH paper which appears to have slowed the use of IVC filters was titled: “Permanent versus Retrievable Inferior Vena Cava Filters: Rethinking the “One-Filter-for-All” Approach to Mechanical Thromboembolic Prophylaxis.”
IVC Filter Dangers Soft Played
IVC filter dangers have been soft played by their manufacturers. The paper’s opening abstract notes: “Deciding which IVC filters to use, when to use them, and for how long they should remain in place remains a highly complex process with many variables to consider. At a minimum, it is important to recognize that mechanical thromboembolic prophylaxis is not entirely benign.” That’s fancy language for admitting that IVC filters can be dangerous.
IVC Filter Dangers Increase over Time
The NIH noted that the use of IVC filters, especially retrievable filters, “is associated with morbidity risks that increase over time.” In plain language, that means the longer an IVC filters remains in the human body, the more likely it is to cause problems. Pieces of the delicate little thimble-sized devices can break off and migrate throughout the circulatory system, including into the lungs or heart. The entire IVC filter can also tilt sideways or embed into a blood vein wall, causing other serious problems, including death.
NIH: Remove Filter after PE Risk Resolves
NIH cautions that IVC “filters should be removed when the patient’s risk for PE has resolved.” The problem is that many temporary filters are being left inside of a person far longer than they should be. To further complicate matters, many IVC filter patients are not told by their doctors that their temporary filters should be removed after a given time. In addition, as many IVC filter lawsuits charge, IVC filter makers such as Bard and Cook Medical have not given their doctor customers clear instructions for when the filters should be removed.
The NIH says that, “Postplacement clinical follow-up is critical to optimizing retrieval rates. Given the increased adverse effects and cost difference when using IVCF in a permanent manner, an important goal for appropriate IVC filter selection should be selecting those patients best served with a permanent filter due to the patient’s unique clinical conditions. . .”
that, “Postplacement clinical follow-up is critical to optimizing retrieval rates. Given the increased adverse effects and cost difference when using IVCF in a permanent manner, an important goal for appropriate IVC filter selection should be selecting those patients best served with a permanent filter due to the patient’s unique clinical conditions. . .”
NIH also recommends further development and validation of nuanced, large-scale mathematical modeling tools to determine how best to use IVC filters. The NIH never directly criticizes any medical device manufacturer, but the language makes it clear that current practices fail IVC filter implantees, or are at least inadequate.
The paper also notes that IVC filter implantation rates remain high compared to rates in Europe.
IVC Filter Background
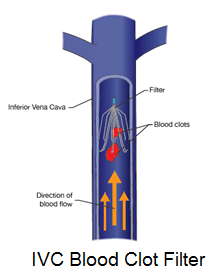
IVC filters are used to prevent deep vein thrombosis (DVT), a medical condition in which blood clots develop in the deep veins of the body, often in the legs, thigh, or pelvis. These clots can break loose and travel to the lungs and can cause a life-threatening condition called pulmonary embolism (PE). IVC filters are implanted in people at risk for PE when anticoagulant therapy is ineffective or cannot be used. They are supposed to be use only in those for whom anticoagulant therapy is contraindicated.
IVC Filter Safety, Efficacy Questioned
IVC filter use has increased rapidly over the years, despite no real evidence that the filters are necessary or effective. In 2010, the U.S. FDA issued a device safety communication after reviewing more than 900 adverse filter events over a five-year period. Those problems include device migration, embolizations, perforation of the IVC, filter fractures. Some led to adverse clinical outcomes, often with the filter remaining in the body long after the risk of PE had subsided. The FDA safety communication recommended filter removal as soon as protection from PE was no longer needed.
 Related
Related
- No Evidence Blood Clot Filters Help
- Texas IVC Filter Lawsuit Filed
- Bard IVC Filter Lawsuit
- Cook IVC Filter Attorney
- IVC Filters poor choice for Trauma Victims

by Matthews & Associates