Mary Nelk brought the complaint (PDF) on February 28, 2023 in the U.S. District Court for the District of New Jersey. Defendant Bard’s headquarters is in the garden state. The lawsuit alleges Ms. Nelk suffered severe injuries from a Bard PowerPort ClearVue Implantable Port. The product was made and sold by Becton Dickinson and Company, as well as its subsidiaries, C.R. Bard and Bard Access Systems, Inc.
Ms. Nelk’s lawsuit alleges that design defects increase the risk of Bard PowerPort problems. The product’s catheter tube is made of a material prone to fracture which can lead to infections, thromboembolism, punctured blood vessels and organs.
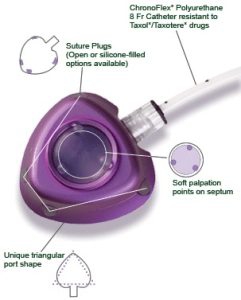
Bard PowerPort at a Glance:
- The PowerPort is implanted below the skin to provide a catheter port for delivery of medications
- The catheter tube is made of a flexible polyurethane polymer called Chronoflex
- The Bard PowerPort lawsuit alleges the tube is prone to cracking, infections, other problems
- Bard PowerPort’s makers knew of fracture risks, yet failed to adequately warn users and medical personnel
Chronoflex AL Design Defect
A product called “Chronoflex,” the lawsuit alleges, is a primary problem in what the suit says is a design defect that leads to the PowerPort’s fracture problems.
Chronoflex AL is one of a large number of biomaterials manufactured by dvanSource.
According to the petition:
“The Chronoflex catheter included in Defendants’ PowerPort is comprised of a polymeric mixture of polyurethane and barium sulfate, a compound which is visible in certain radiologic studies.
Barium sulfate is known to contribute to reduction of the mechanical integrity of polyurethane in vivo as the particles of barium sulfate dissociate from the surface of the catheter over time, leaving microfractures and other alterations of the polymeric structure and degrading the mechanical properties of the catheter.
The mechanical integrity of a barium sulfate-impregnated polyurethane is affected
by the concentration of barium sulfate as well as the homogeneity of the modified polymer.”
The petition alleges that Defendants’ Chronoflex Catheter manufacturing process involved a too-high concentration of barium sulfate particles. That concentration allegedly led to improperly high viscosity of the raw polyurethane before polymerization, causing improper mixing of barium sulfate particles within the polymer matrix.
That improper mixing led to pockets of barium sulfate and entrapped air being distributed through the catheter body, as well as on its inner and outer surfaces.
The petition further states:
“This defect in the manufacturing process led to a heterogeneous modified polymer which led to an irregular catheter surface replete with fissures, pits and cracks. The roughened catheter surface leads to the collection and proliferation of fibrinous blood products, thereby drastically increasing the risk of thromboembolism.
Although the surface degradation and resulting risk of thromboembolism can be reduced or avoided with design modifications to encapsulate the radiopaque compound or by using a different polymer formulation, Defendants elected not to incorporate those design elements into the PowerPort.”
Petition: PowerPort System not Safe and Effective as promoted
The defendants misrepresented the safety of the PowerPort system, says the petition. They negligently designed, manufactured, prepared, compounded, assembled, processed, labeled, marketed, distributed, and sold the PowerPort system as safe and effective when it was not.
The petition further alleges that Bard PowerPort’s defective polymer design increases risk of catheter bloodstream infections, blood clots, and punctured blood vessels.
What is the Bard PowerPort?
The Bard PowerPort is a vascular access device that is implanted below the skin. It is designed to provide a catheter port which allows easy access to a patient’s bloodstream in order to deliver medications. The device consists of two parts: 1) an injection port through which a needle is inserted for medications; 2) a polyurethane catheter tube which carries drugs into the blood vessel.
Missouri Woman Mary Nelk
According to her Bard PowerPort lawsuit, Ms. Nelk was implanted with the device in her internal jugular vein during February 2021 in order to receive nutrient treatments for Crohn’s disease. Days later, she presented at hospital with arm pain, hematoma, high fever. Doctors diagnosed her with a bloodstream infection, then discovered her Bard PowerPort was not working.
Those Bard PowerPort problems led Ms. Nelk to multiple hospital admissions for infections. She said she now suffers severe and permanent injuries from those infections.
Bard PowerPort Catheter Problems
Becton Dickinson and its Bard units have known for years that there was a problem with the Bard PowerPort’s catheter tubes, according to the complaint, which also alleges that the tubes are prone to fissuring and cracking, which can lead to infections, punctured blood vessels, thromboembolism.
Bard ignored Large Numbers of Adverse Events Reports
Ms. Nelk’s petition also states that Defendants began receiving large numbers of adverse events reports (AERs) years before she was implanted with the device. Those reports from health care providers indicated that the PowerPort was precipitating thromboembolism post-implantation. In addition, Defendants also received many AERs reports that said PowerPort perforated internal vasculature.
AERS of Injuries included:
- Hemorrhage
- Cardiac/pericardial tamponade
- Cardiac arrhythmia
- Severe and persistent pain
- Perforations of tissues, vessels and organs
- Patient death
Defendants Failed to Alter Defective Design – Petition
Ms. Nelk’s lawsuit also says that the PowerPort makers did not alter the design to make it safer as they should have done. The defendants chose instead to “actively and aggressively market the PowerPort as safe, despite their knowledge of numerous reports of thromboembolism and other serious injuries.”
Causes of action in the lawsuit include:
- Negligence
- failure to warn
- manufacturing defect
- design defect
- breach of warranty
- fraudulent concealment
- violations of the Missouri Merchandising Practices Act and the New Jersey Consumer Fraud Act.
The Missouri woman seeks both compensatory and punitive damages in unspecified dollar amounts.
RELATED
- Bard PowerPort Lawsuit | Lawyer
- Free Legal Consultation

by Matthews & Associates