 The US Food & Drug Administration has recalled at least 19 medical devices so far in 2021. What follows is a sampling of seven medical devices the FDA has recalled as of May 21.
The US Food & Drug Administration has recalled at least 19 medical devices so far in 2021. What follows is a sampling of seven medical devices the FDA has recalled as of May 21.
• Penumbra Catheter
Penumbra recalled the Penumbra JET 7 Xtra Flex in December 2020 because the catheter may become susceptible to distal tip damage during use. The FDA had received more than 200 medical device reports (MDRs) associated with the JET 7 Xtra Flex catheter, including deaths, serious injuries, and malfunctions. Twenty of these MDRs describe 14 unique patient deaths, which include reports from different reporting sources for a single adverse event. Other MDRs describe serious patient injury such as vessel damage, hemorrhage, and cerebral infarction. Device failure modes reported in the MDRs include ballooning, expansion, rupture, breakage, or complete separation, and exposure of internal support coils near the distal tip region of the JET 7 Xtra Flex catheter.
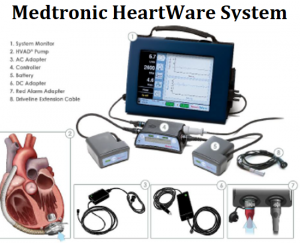
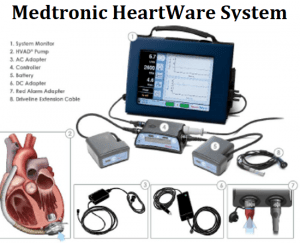
• Medtronic HeartWare System
Medtronic recalled its HeartWare HVAD System to provide updated Instructions for Use (IFU) and Patient Manual (PM) due to safety issues with (1) Carrying Cases, (2) Driveline Cover Orientation and; (3) Controller Power-Up Sequence.
- Carrying Case: A drop of the carrying case, caused by damage to the case or improper wear, can disconnect the driveline and interrupt pump power.
- Driveline Cover Orientation: If the driveline cover is first installed properly during the surgical implant procedure, it will always cover the driveline connector. However, if a patient removes the cover during a controller exchange, there is a possibility that the driveline cover could be put on backwards. In that reversed position, the fit of the cover inadvertently causes the driveline locking mechanism to be in the unlocked position, which could cause temporary or accidental driveline disconnects.
- Controller Power-Up Sequence: During power-up, the LED lights turn red and can be misunderstood as a “red alarm,” leading to an unnecessary controller exchange. The IFU and PM will be updated to make clear the expected power-up sequence causes the alarm indicator LEDs and both sets of battery LEDs to turn red for 2.5 seconds while the controller LCD displays the power-on message.
If using the HVAD system and (1) the carrying case breaks and the driveline pulls out of the controller as it drops, or (2) the driveline disconnects from backwards driveline cover orientation; or (3) a controller exchange is performed unnecessarily due to confusion of start-up behavior as a “red alarm” battery failure; this may cause serious patient harm, including death.
One death and 64 injuries have been reported to the FDA for these issues.
• Boston ScientificEMBLEM S-ICD Electrode
Boston Scientific recalled the EMBLEM S-ICD Subcutaneous Electrode because of increased risk of fractures at a specific point distal to the proximal sense ring. If the device fractures during use, it could become unable to deliver therapy to slow very fast heartbeats from cardiac arrest (tachycardia). A failed device may cause serious adverse events. Examples include injury or death if cardiac arrest cannot be treated or need for additional surgery to replace failed devices.
There have been 27 complaints about this device issue and 26 reports of serious injuries. One death has been reported
• Cordis Carotid System
Cordis Corporation announced in February that it was recalling its Precise PRO Rx US Carotid System due to the risk of separation in the medical device.
The Precise PRO Rx US Carotid System is used to treat patients with narrowed carotid arteries. The system includes a metal (nitinol) self-expanding stent preloaded on a delivery catheter used to place the stent.
The FDA identified this recall as its most serious type: a Class 1 recall. This means the agency has determined that using these devices could cause serious injuries or death.
To date, there have been seven complaints over the recalled device, including five reported injuries, though no deaths have been reported.
• Boston Scientific Stent
Boston Scientific is recalling the VICI SDS and RDS VENOUS STENT Systems after reports indicate that the stents may migrate or move from where they are initially implanted.
A migrated stent may require another surgery or catheter procedure to retrieve it, which increases risks to the patient, including possible damage to the blood vessel, heart walls, or other organs. If the stent migrates to the heart, it could cause life-threatening injury.
There have been 17 complaints and reported injuries related to this issue, though no deaths have been reported.
• Abbott Assurity™ and Endurity™ Pacemakers
Abbott (formally “St. Jude Medical”) is recalling a subset of Assurity and Endurity pacemakers built using specific manufacturing equipment, that were then distributed from April 2015 to February 2019. A small number of devices from that time frame have experienced problems when moisture is able to get inside the device and potentially cause an electrical short that may lead to:
- A loss of device pacing
- Telemetry failure or errors in information
- Early and fast battery drain
- Less time between the first battery depletion warning (elective replacement indicator or ERI) and the device’s end of service (EOS)
If the device is unable to deliver pacing, patients may experience slow or irregular heartbeat, fainting, shortness of breath, tiredness, dizziness, or discomfort. Additionally, shorter battery life and device life may lead to an additional pacemaker replacement procedure sooner than expected. Finally, if the system does not relay accurate information via telemetry, medical providers may not know to provide treatment.
There have been 135 complaints, 135 injuries, and no deaths reported for this issue.
• Medtronic HVAD Pump Implant Kit
Medtronic recalled its HVAD Pump Implant Kit because the device may fail to initially start, restart, or have a delay in restarting after the pump was stopped. These delays or failures to start or restart have occurred during preimplant testing, during the implant, or in a variety of post-implant situations. If the device delays or fails to start or restart, serious patient harm could result, including a heart attack, worsening heart failure, the need for additional procedures and hospitalizations, or death.
There have been 29 complaints that include two deaths, 19 serious injuries and 8 cases of patients who had a life-threatening event but recovered without long term effects.
RELATED
- 2021 FDA Medical Device Recalls
- Medical Devices Wild West for Profits, Problems
- IVC Filter Lawsuit | Lawyer

by Matthews & Associates