 Abbott announced Feb. 17, 2022 that it was recalling some powder infant formulas following four consumer complaints that included the death of an infant. Abbott recalled powder infant formulas Similac, Alimentum, and EleCare.
Abbott announced Feb. 17, 2022 that it was recalling some powder infant formulas following four consumer complaints that included the death of an infant. Abbott recalled powder infant formulas Similac, Alimentum, and EleCare.The recalled powder formula products were all made in Abbott’s manufacturing facilities in Sturgis, Michigan.
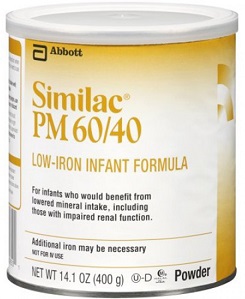
On Feb. 28, federal officials reported that Abbott Nutrition had widened its recall of the formula. That report came as the FDA said it was probing another fatal case of Cronobacter sakazakii infection involving an infant fed the Abbott Similac PM 60/40 formula with a lot code of 27032K800.
“This is a specialty formula for certain infants who would benefit from lowered mineral intake and was not included in the previous recall,” FDA officials said in a statement.
The FDA announced Feb. 17 that Abbott was voluntarily recalling certain lots of its Similac, Alimentum and EleCare formulas after four infants were sickened, including one who died.
“All five illnesses resulted in hospitalization, and Cronobacter may have contributed to death in two patients,” said FDA officials. The cases were reported in Minnesota, Ohio and Texas.
Cronobacter sakazakii
Four reports of Cronobacter sakazakii — a rare bacteria that can cause several different types of infections, including severe meningitis in infants — and one report of Salmonella Newport infection have been linked to the recalled formulas.
Statement from Abbott
Abbott announced last month: “[As] part of Abbott’s quality processes, we conduct routine testing for Cronobacter sakazakii and other pathogens in our manufacturing facilities. During testing in our Sturgis, Mich., facility, we found evidence of Cronobacter sakazakii in the plant in non-product contact areas. We found no evidence of Salmonella Newport. This investigation is ongoing.”
Abbott further said that it conducts extensive quality checks on each completed batch of infant formula, including microbiological analysis prior to release:
“All finished products are tested for Cronobacter sakazakii, Salmonella Newport and other pathogens and they must test negative before any product is released. Additionally, retained samples related to the three complaints for Cronobacter sakazakii tested negative for Cronobacter sakazakii. And the retained sample related to the complaint for Salmonella Newport tested negative for Salmonella Newport.”
Cronobacter sakazakii is commonly found in the environment and some areas in the home. It can cause fever, poor feeding, excessive crying, low energy, other serious symptoms. It’s important to follow the instructions for proper preparation, handling and storage of powder formulas.
RELATED
- Toxic Baby Food Lawsuit
- Infant Formula Lawsuit | Necrotizing Enereocolitis (NEC)
- FDA Press Announcement
- FDA Advisory

by Matthews & Associates