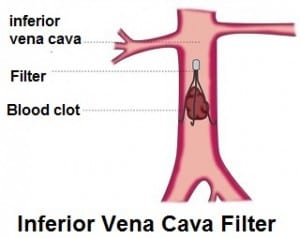
 A jury last week awarded a woman $3.3 million in a case that stemmed from her injury caused by an IVC blood clot filter. The jury unanimously made the award in a Wisconsin courtroom. A woman, Natalie J., filed the lawsuit after she was implanted with C.R. Bard’s Meridian IVC Filter prior to undergoing surgery for varicose veins in the legs.
A jury last week awarded a woman $3.3 million in a case that stemmed from her injury caused by an IVC blood clot filter. The jury unanimously made the award in a Wisconsin courtroom. A woman, Natalie J., filed the lawsuit after she was implanted with C.R. Bard’s Meridian IVC Filter prior to undergoing surgery for varicose veins in the legs.C.R. Bard promotes the IVC filter as a medical device made to help prevent blood clots from developing in the legs and traveling to the lungs, which can result in a life-threatening pulmonary embolism. This is exactly what happened to the patient.
The Meridian IVC filter caused the woman’s life-threatening complications. The filter tilted and punctured a vein just days after doctors implanted it in her vein.
A 2018 CT scan revealed that the woman’s IVC Filter had broken into pieces, with one of the pieces traveling to her heart. Another dislodged piece migrated and embedded into her vein.
These life-threatening complications led to multiple surgeries that were needed to remove the filter and the broken pieces.
The $3.3 million verdict in Wisconsin was the latest in a string of several victories for plaintiffs’ lawyers in IVC filter cases in the last three months. The seven-figure verdict in Wisconsin followed a $926,000 verdict awarded by an Oregon jury, in addition to a $2.5 million verdict awarded a few weeks earlier by a Texas jury in Dallas.
IVC Filter History
IVC filters were allegedly designed to stop blood clots. The problem is that, as happened in the Wisconsin woman’s case, they can break off and migrate to the heart, lungs, or other vital organs. IVC filters can cause fatal injuries. The medical literature shows a significant statistical rate of IVC (Inferior Vena Cava) filter failure, fracture, device migration, embolization (movement of the filter or fractured fragments to the heart or lungs), perforation of the IVC, and other problems.
Other IVC Filter Verdicts
Other IVC filter verdicts for plaintiffs include a November 2019 trial in which a jury awarded a $34 Million jury verdict against Rex Medical for a Georgia woman injured by a Rex Option IVC vein filter.
IVC Filters Show No Evidence of Benefit
The Journal of the American Medical Association (JAMA) has reported that IVC blood clot filters show evidence of harm with no evidence of benefit. Bard Recovery IVC filters may even be fatally flawed.
FDA Warning: Filters Short-Term Only
On May 6, 2014, the FDA updated its safety communication: “Removing Retrievable Inferior Vena Cava Filters.” The intended audience was implanting physicians and clinicians in charge of caring for patients with inferior vena cava (IVC) filters, as well as interventional radiologists and cardiologists, vascular surgeons, emergency room (trauma) and primary care physicians, bariatric and orthopedic surgeons.
In August 2010, FDA issued an initial warning against leaving inferior vena cava filters implanted in patients for extended periods due to their potential to cause adverse health complications. The FDA warned that IVC filters are meant for short-term use only, and only for patients at risk for pulmonary embolism. FDA advised implanting doctors to remove the devices once PE risk subsides. The agency was concerned about doctors not retrieving IVC filters intended only for short-term placement, hence exposing patients to fractures and all the problems which can follow.
FDA Reports IVCF Adverse Events
Since 2005, the FDA said it has received “921 device adverse event reports involving IVC filters, 328 for device migration; 146 for embolizations (detachment of device components); 70 for perforation of the IVC; 56 for filter fracture.” Some of these events led to problems for patients, according to FDA, which also said the events could be related to a retrievable filter remaining in patients’ bodies beyond the time when pulmonary embolism (PE) risk subsided:
“The FDA is concerned that these retrievable IVC filters, intended for short-term placement, are not always removed once a patient’s risk for PE subsides. Known long term risks associated with IVC filters include but are not limited to lower limb deep vein thrombosis (DVT), filter fracture, filter migration, filter embolization and IVC perforation.”
RELATED
- IVC Filter Lawsuit | Attorney
- Cook Medical loses $1.2 Million IVC Filter Verdict
- IVC Filters overused, unproven
- IVC Filters poor choice for Trauma Victims
- New Study questions IVC Filter Safety, Uses

by Matthews & Associates