IVC Filter Lawsuit Lawyer
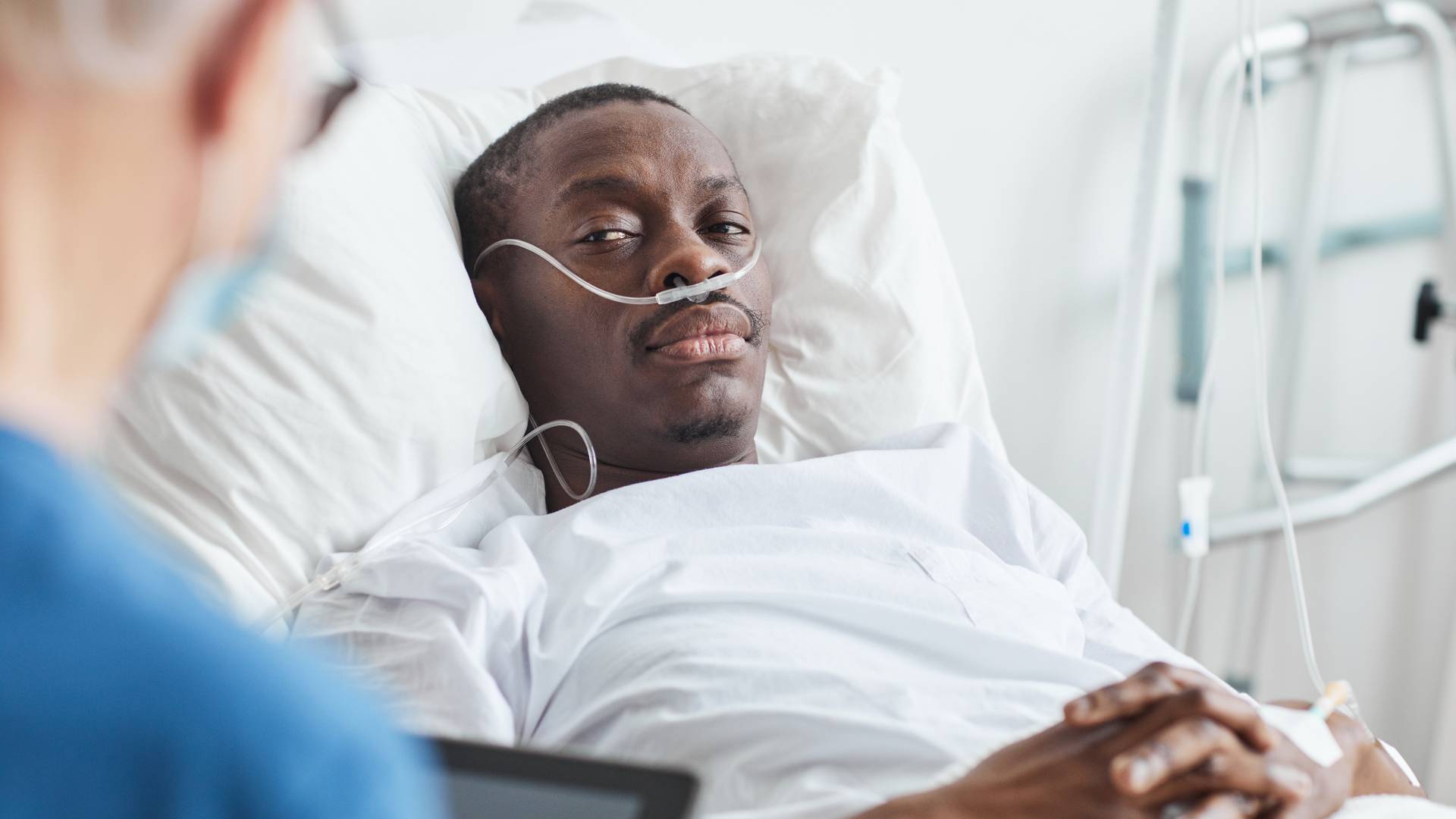
IVC filters, allegedly designed to stop blood clots, can break off and migrate into the heart, lungs, and other vital organs. IVC filters can cause fatal injuries. The medical literature shows a significant statistical rate of IVC – Inferior Vena Cava – filter failure, fracture, device migration, embolization (movement of the filter or fractured fragments to the heart or lungs), perforation of the IVC, and other problems.
If you have been injured by a blood clot filter, or had an IVC filter installed which migrated and could not or cannot be safely removed, contact an experienced IVC Filter Lawyer for a potential Bard IVC Filter Lawsuit, or a Bard Recovery Blood Clot Filter Lawsuit. Please view our testimonials to see how we have helped others injured by dangerous medical devices and pharmaceutical drugs.
IVC Filter Litigation Update:
- (Nov. 27, 2019) Matthews & Associates won a large jury verdict against Rex Medical for a Georgia woman injured by a Rex Option IVC vein filter.
- Matthews & Associates won a large jury verdict in a Texas courtroom in May 2018 for a Houston-area firefighter. Represented by David Matthews as well as Freese & Goss, Jeff Pavlock was awarded damages against Cook Medical over an IVC filter.
IVC Filter Litigation
IVC filters most often seen in the litigation include the Bard Recovery filter, Bard G2 filter, Bard G2 Express filter, Cook Gunther Tulip filter, Cook Celect filter, and Rex Medical Option filter. Other troublesome IVC blood clot filters include the Bard Eclipse, Bard Meridian, Bard Denali IVC.
Our law firm is filing Bard IVC Filter Lawsuits and Cook IVC Filter Lawsuits nationwide. Contact us by calling (888) 923-7001 for a free legal consultation.

We may file or settle a claim individually or as part of a group on a case-by-case basis.
Whether your claim is filed individually or as part of a group fighting the same defendant, our goal is always to provide each of our clients with individual attention. This boutique-level service, combined with our national litigation powerhouse capabilities, ensures you receive both personalized care and the resources necessary to take on the largest corporations and manufacturers.
No Evidence of Benefit for IVC Filters
The Journal of the American Medical Association (JAMA) has reported that IVC blood clot filters show evidence of harm with no evidence of benefit. Bard Recovery IVC filters may even be fatally flawed. An IVC Filter Lawsuit Lawyer may be able to help with a free legal consultation if you or someone you know has been implanted with an IVC filter.
FDA Reports IVC Blood Clot Filter Injuries
The FDA updated a 2010 safety communication in May 2014 regarding device removal complications. FDA has received more than 1,000 reports of serious adverse events related to Bard IVC Filters. Reports include 328 for device migration, 146 for embolizations (device component detachment), 70 for perforation of the IVC, and 56 for filter fracture. Similar problems have been found with other IVC filters made by Cook Medical, Boston Scientific, B. Braun and others.
What Is the Vena Cava?
The vena cava is a large vein that carries deoxygenated blood into the heart. Two vena cava transport blood through human beings: the inferior vena cava carries blood from the lower body; the superior vena cava moves blood from the head, arms and upper body.
IVC Filter Profile
Inferior Vena Cava filters are tiny umbrella skein-like devices inserted into the inferior vena cava to capture blood clots and prevent them from reaching the lungs or heart. IVC filters are often put in patients at risk for pulmonary embolism (PE, or a blood clot in the lungs) when anticoagulant therapy is ineffective or contraindicated. If patients require long-term protection from PE, surgeons may implant permanent IVC filters. If people require short-term protection, doctors may implant retrievable IVC filters. In theory, the latter are designed to allow device removal once PE risk subsides.
Bard IVC Filter Studies
A research team led by Dr. William Nicholson at York Hospital in Pennsylvania examined 80 patients who had been implanted with Bard IVC filters from 2004 – 2009. The 80 underwent a fluoroscopy to determine the structural integrity of the filter and investigate whether it had broken or was still intact. Patients whose devices were fragmented also underwent echocardiography and cardiac computed tomography.
IVC Filter Fractures
The study’s results indicated that 16% – 13 of 80 patients – who received Bard Recovery and Bard G2 filters had at least one fracture. First-generation Bard Recovery filters had a 25% (7 of 28) fracture rate, with embolizations to the heart occurring in five (5) cases. Of second-generation devices (including the Bard G2 filter), 12% – 6 of 52 – were found to have fractured. In two cases, the fragment inhibited blood flow, one in the vein coming out of the liver and one in the lungs. In the other four cases, the fragments stayed close to the filter.





Bard IVC Filter Litigation
In September 2012, a class action lawsuit filed against C.R. Bard over its Recovery and G2 IVC filters was removed from Florida state court to the federal court system. Attorneys for Bard filed a petition to have the case transferred to the U.S. District Court for the Southern District of Florida, which means that the company believes the class action will include more than 100 plaintiffs; that total claims could top $5 million; that members of the plaintiff class will be diverse from at least one defendant.
Many other Bard IVC filter lawsuits have been filed in Pennsylvania and California state courts. Each involves allegations that Bard knew – or should have known – about its products’ potentially serious side effects yet failed to warn of them. The claims seek compensation for the medical monitoring patients will need to ensure their IVC filters have not fractured or embolized when they remain implanted. Some 100,000 Bard IVC filters have been implanted in patients around the country.
Cook Medical MDL Set in Indiana
In October 15, 2014, lawsuits involving inferior vena cava (IVC) filters made by Cook Medical were centralized in a federal Multi-District Litigation (MDL) court in Indiana.
The Judicial Panel on Multi-District Litigation (JPML) issued an order (PDF) that centralizes 27 lawsuits pending in eleven district courts into the U.S. District Court for the Southern District of Indiana. Federal judges selected Indiana because half the lawsuits against Cook have already been filed there. Cook Medical headquarters is also in Bloomington.
JPML courts help eliminate duplicative discovery, prevent inconsistent pretrial rulings, and conserve the resources of all parties, their attorneys and the judiciary.
Cook IVC Filter Lawsuits: Günther Tulip & Celect
Cook Medical is accused of selling defective IVC filters and failing to sufficiently warn of safety risks. Most of the lawsuits involve the Günther Tulip and Celect filters.
According to the JPML: “The subject actions share factual issues arising from allegations that defects in the design of Cook’s inferior vena cava (“IVC”) filters makes them more likely to fracture, migrate, tilt, or perforate the inferior vena cava, causing injury.”
Cook Medical opposed centralization in an MDL. Its lawyers argued centralization would encourage plaintiffs to file meritless claims. Cook's attorneys argued, “perforation and fracture are extremely rare occurrences.” They added that they will seek dismissal of claims by arguing that doctors were adequately warned about risks. This tactic is known as the “learned intermediary defense,” which says the doctor was warned and has the duty to warn the patient, that the company has no obligation to warn the patient.
IVC Filter Complications
Fracture of the IVC Filter can cause:
- Perforation or puncture of the IVC
- Serious damage to the heart
- Damage to the Lungs or Vena Cava
- Internal Bleeding
- Cardiac or Pericardial Tamponade
- Ventricle Tachycardia
- Lower Limb DVT
- G2 X
- G2
- Pulmonary Embolism
- Infection
- Death
- Bard IVC Filters
- Recovery
- Denali
- Meridian
- Eclipse
- Hematoma or Nerve Injury at the Puncture Site
- Constant and Severe Pain in the Heart, Chest, elsewhere
IVC Filter Updates
We will try to keep you up to date on any IVC Filter lawsuits, additional FDA IVC Filter warnings, and IVC Filter recall announcements.
Learn more about how we can help you by contacting us online or calling (888) 923-7001 today!
We're Ready to Fight For You!
-
“Very professional from the beginning”
- Jeffrey E.Very professional from the beginning. always answered questions and had amazing follow through on insuring data submitted was accurate
-
“Matthews and Associates are very informative. Kept me up to date on what was happening with my case.”
- Ron S.Matthews and Associates are very informative. Kept me up to date on what was happening with my case. They were always polite when they were talking to me. Very professional and polite.
-
“Very informative and professional.”
- Lydia C.Very informative and professional. Answer thoroughly all my questions and concerns.
-
“They are so helpful, kind, and knowledgeable.”
I love these guys! They are so helpful, kind, knowledgeable, smart, and good with people. They answer all your questions and if they don't know the answer, it doesn't take them long to get you the answer! I wouldn't want anyone else to handle my suit. They are AWESOME!- Genie S. -
“Matthews & Associates is one of a kind.”
Matthews & Associates is one of a kind. They treat you with respect. They support you when needed. Plus they have locations in Texas and California with wonderful customer service. Bilingual service is always available.- Sorangel T. -
“Always available & answers questions promptly.”
I've been working with Matthews & Associates for 2 years. I am impressed that the firm is always available and answers questions promptly. I am very satisfied with the services they provide and would recommend them.- Kweli Y.
Do I Have a Bard IVC Filter Lawsuit or Blood Clot Filter Lawsuit?
Contact an experienced IVC filter attorney at Matthews & Associates for a free legal consultation if you believe you may have cause to pursue a Bard IVC Filter Lawsuit or a Blood Clot Filter Lawsuit against the makers of a blood clot filter.
Schedule your initial consultation by calling our firm at (888) 923-7001 or filling out our online form.
RELATED:
- Bard Recovery Blood Clot Filter Lawsuit
- Medical Devices not tested for Safety
- Blood Clot Filter Case filed in Missouri
- No Evidence Blood Clot Filters help
- Cook Blood Clot Filter Lawsuits
- IVC Filter MDL Court for Cook Medical
- IVC Filter Patients Unaware of Implants
- Retrievable IVC Filters not retrieved
- IVC Filter Problems Unreported
- IVC Filter Controversy