
Philips CPAP Recall Lawsuit
Philips Respironics has issued a recall for millions of its DreamStation CPAP sleep apnea and other breathing machines due to an increased risk of people developing severe side effects from using these products. The devices are being recalled over concerns that their foam liners may disintegrate or offgas, potentially causing users to inhale contaminants and/or toxic chemicals. Anyone who has experienced severe side effects after using a Phillips Bi-Level Positive Airway Pressure (Bi-Level PAP), Continuous Positive Airway Pressure (CPAP), or mechanical ventilator device may be eligible for compensation by filing a Philips CPAP lawsuit.
Philips CPAP Recall Update: 3-4 Million Devices Recalled
On April 26, 2021, Philips provided an important update regarding its efforts to address issues the company identified in several of its Sleep and Respiratory Care products. Further, Philips ceased all sales of its DreamStation 1 CPAP products in the US, then updated that recall last month, in June 2021. Because the recall is considered voluntary, it will likely be considered a Class II FDA recall. It is currently listed as a safety notification.
The recall involves millions of Philips’ sleep apnea and ventilator machines, most of which are of the DreamStation, first-generation production line and are within the 5-year service life. Philips issued the recall for machines that contain a sound-abatement foam comprised of polyester-based polyurethane which may degrade to release particles and chemicals that can be inhaled. Philips estimates that up to four (4) million devices may be affected in the U.S. market.

We may file or settle a claim individually or as part of a group on a case-by-case basis.
Whether your claim is filed individually or as part of a group fighting the same defendant, our goal is always to provide each of our clients with individual attention.
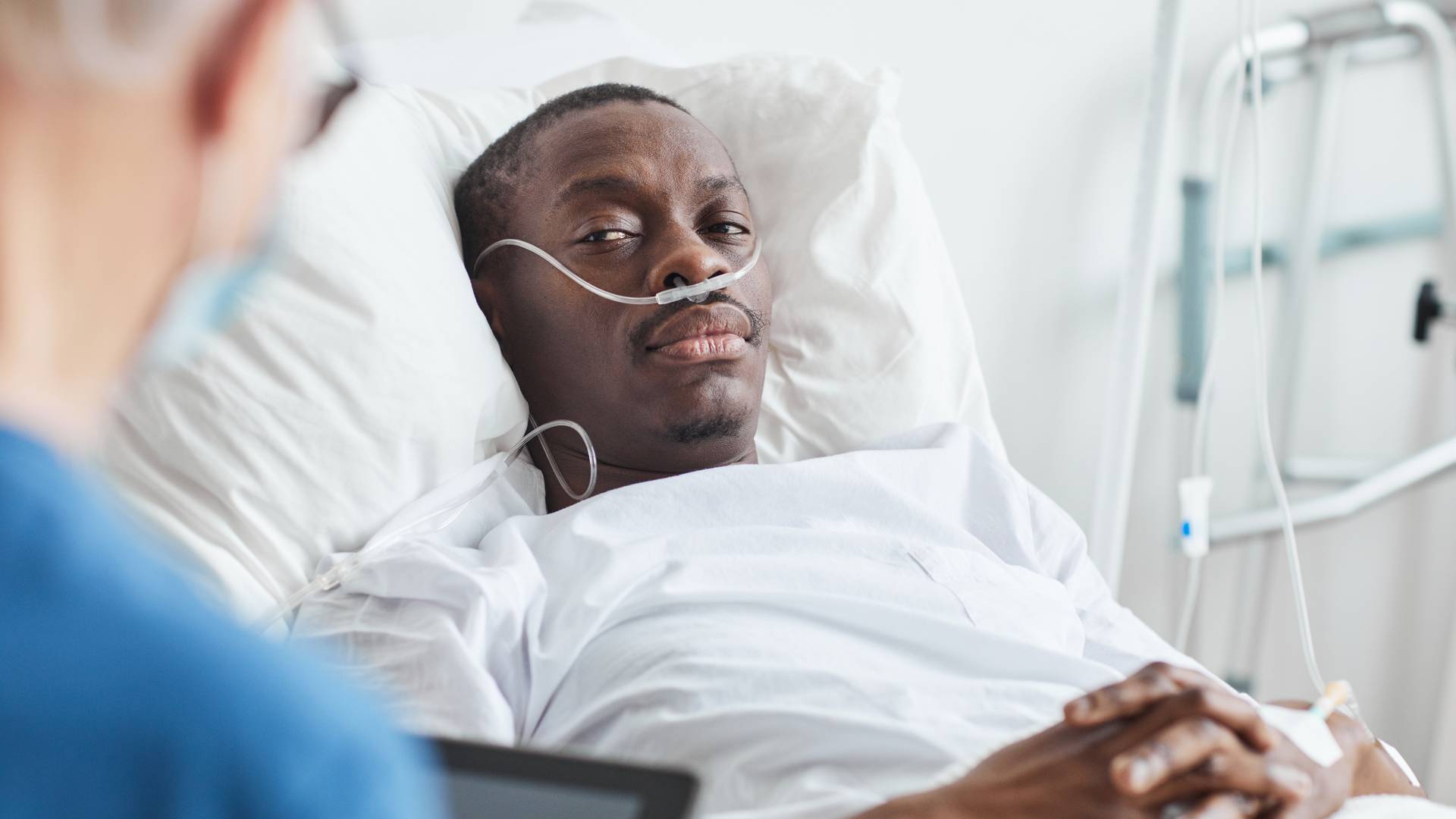
Philips Breathing Machines Symptoms of Exposure
Though actual complaints have been few to date, some users have reported black debris collecting in their breathing tubes. The potential degradation of PE-PUR foam may lead to inhalation of degraded products and toxic chemicals from initial use of the Philips devices. This problem could increase the chance of severe adverse side effects and might also increase cancer risk.
Symptoms of exposure may include:
- Coughing
- Headache
- Throat irritation
- Lung inflammation
- Nausea and vomiting
- Sinus infections
- Carcinogenic effects
- And more
Recalls and Countries Affected
- United States
- Canada
- Australia



Sound-Reducing Foam Liner Suspected
Risks of severe adverse side effects, including cancer, appear to be related to the polyester-based polyurethane (PE-PUR) foam used as a sound-abatement component in certain models of the devices. PE-PUR foam may degrade into particles that could enter into the device air pathway and be inhaled or ingested by the user. The foam may also be prone to “offgas” chemicals that could be toxic. Degradation and chemical offgassing problems could be exacerbated by unapproved cleaning methods that include high heat, high humidity, and ozone cleaning environments.
Canadian government officials have noted: “[R]esults of testing performed by Philips indicate the PE-PUR sound abatement foam used in these devices may emit certain chemicals. Our investigation to date indicates that this emission occurs during initial operation and may possibly continue throughout the device’s useful life.”
Philips Respironics notes that the absence of visible particles does not mean that foam breakdown has not already begun. Lab analysis of the degraded foam reveals the presence of potentially harmful chemicals that include:
• Toluene Diamine
• Toluene Diisocyanate
• Diethylene glycol
Lab testing performed for and by Philips also identified the presence of VOCs which may be emitted from the sound abatement foam component of the affected device(s). VOCs are emitted as gases from the foam included in the CPAP, Bi-Level PAP, and mechanical ventilator devices. Such gases may have short- and long-term adverse health effects. Philips’ own lab testing identified two compounds of concern that may be emitted from the foam that are outside of safety thresholds: Dimethyl Diazine and Phenol, 2,6-bis (1,1-dimethylethyl)-4-(1-methylpropyl).
Philips Recall Lawsuits
Philips may soon face several lawsuits filed by people who were diagnosed with cancer or other injuries (listed below) after using a Philips Sleep & Respiratory Care CPAP, Bi-level PAP, or a mechanical ventilator.
Philips has provided a “Sleep and Respiratory Care update: Clinical information for physicians.” It lists the following possible Injuries Philips device users could suffer from inhaling or ingesting foam particles:
Potential risks of degraded foam exposure include:
- Headache
- Inflammatory response
- Irritation (skin, eyes, and respiratory tract)
- Asthma
- Adverse effects to other organs (e.g. kidneys and liver)
- Toxic and carcinogenic effects (cancers)
Potential Risks from Volatile Organic Compounds (VOCs) from degraded foam exposure include:
- Irritation (skin, eyes, nose, respiratory tract)
- Headache/dizziness
- Hypersensitivity
- Nausea/vomiting
- Toxic and carcinogenic effects (cancer)
-
“Matthews and Associates are very informative. Kept me up to date on what was happening with my case.”
- Ron S.Matthews and Associates are very informative. Kept me up to date on what was happening with my case. They were always polite when they were talking to me. Very professional and polite.
-
“I am impressed that the Firm is always available and answers questions promptly”
- Kweli Y.I've been working with Matthews and Associates for 2 years. I am impressed that the Firm is always available and answers questions promptly. I am very satisfied with the services they provide and would recommend them.
-
“I Love these guys!!!! They are SO Helpful, Kind, Knowledgeable,”
I Love these guys!!!! They are SO Helpful, Kind, Knowledgeable, Smart, good with people, answer ALL ur questions, if they don't know the answer, it sure doesn't take them long to get you the answer!!!!!!!!!! I wouldn't want anyone else to handle my suit. They are AWESOME!!!!!!!!!- Genie S. -
“Very informative and professional.”
- Lydia C.Very informative and professional. Answer thoroughly all my questions and concerns.
-
“They treat you with respect. They support you when needed.”
- Sorangel T.Matthews and Associates is one of a kind. They treat you with respect. They support you when needed. Plus they have locations in Texas and California with wonderful customer service. Billingual service always available.
-
“Very professional from the beginning”
- Jeffrey E.Very professional from the beginning. always answered questions and had amazing follow through on insuring data submitted was accurate
About Philips Group
Philips is one of the largest makers of sleep apnea machines and ventilators in the world. An estimated 3-4 million devices are targeted in this recall. Philips Group company spokesman Steve Klink said that approximately 80% of the affected devices in the recall were machines used to help people with sleep apnea, known as Continuous Positive Airway Pressure (CPAP) machines. All users of those machines were advised to halt usage. About two-thirds of Philips CPAP machine sales occur in the United States. The other 20% of affected devices were mechanical ventilators.
Philips’ 2020 Annual Report indicates Philips Group generated approximately EUR 19.5 billion ($23.14 billion) in total sales. Philips’ “Connected Care” business segment (which contains Philips’ Sleep & Respiratory Care Products) comprised roughly 28% of total sales – approximately $6.6 billion US dollars or EUR 5.564 billion. Philips Group CEO Frans van Houten announced that the company has set aside EUR 500 million ($606 million) to date for dealing with this recall. The company stated in its Q1 Earnings Call that this amount does not account for potential litigation that may ensue.

Sources:
- Philips issues recall notification to mitigate potential health risks related to the sound abatement foam component in certain sleep and respiratory care devices, Philips (June 14, 2021)
- URGENT: Medical Device Recall: Sound Abatement Foam Susceptibility to Degradation and Volatile Organic Compound Emission, Philips Respironics (June 2021)
- Sleep and Respiratory Care update, Clinical information for physicians, Philips (June 14, 2021)
- Kassraie, Aaron, Philips Recalls Millions of Sleep Apnea CPAP Machines, Ventilators, AARP (June 15, 2021)
- Gibson, Kate, Philips recalls ventilators and sleep apnea CPAP machines over cancer concerns, CBS News (June 15, 2021)
- Continuous Positive Airway Pressure (CPAP), Bi-Level Positive Airway Pressure (BiPAP), and Mechanical Ventilators, Government of Canada (June 23, 2021)
- Philips recall action for CPAP, Bi-Level PAP devices and mechanical ventilators: Safety hazard caused by foam degradation and emissions, Australian Government: Department of Health (June 25, 2021)
- Sterling, Toby, Philips recalls ventilators, sleep apnea machines due to health risks, Reuters (June 14, 2021)
- Philips Annual Report 2020 (February 23, 2021)
- Philips Q1 2021 Earnings Call Transcript, Motley Fool (April 26, 2021)


