 Jane Akre of Mesh Medical Device News Desk reports from Missouri that Dr. Anne Weber testified this week on day four of the trial of Budke v. Ethicon (Johnson & Johnson). Family survivors of Joan Budke claim Johnson & Johnson’s Prolift pelvic mesh implant led to her death 16 months after the experimental plastic contraption was sewn inside her. Ms. Budke died from massive infections which plaintiffs say were both caused and exacerbated by the plastic mesh.
Jane Akre of Mesh Medical Device News Desk reports from Missouri that Dr. Anne Weber testified this week on day four of the trial of Budke v. Ethicon (Johnson & Johnson). Family survivors of Joan Budke claim Johnson & Johnson’s Prolift pelvic mesh implant led to her death 16 months after the experimental plastic contraption was sewn inside her. Ms. Budke died from massive infections which plaintiffs say were both caused and exacerbated by the plastic mesh.
Camden Co. near Lake of the Ozarks, Mo. – where Ms. Budke lived – is the site of the trial. Her husband Donald and the three adult Budke children brought the lawsuit. Two daughters were in the courtroom when Dr. Anne Weber testified.
Dr. Anne Weber testifies
Dr. Weber was called to the stand by plaintiffs on Jan. 12. She is a transvaginal mesh expert who did a residency in obstetrics and gynecology and was also an advanced surgical resident at the Cleveland Clinic. Dr. Weber was an originator of the specialty of urogynecology. She worked and trained others in the field at the world-renowned Cleveland Clinic. Students, residents and fellows at the clinic train in the specialty that becomes urogynecology after a four-year residency in Obstetrics/Gynecology.
At the National Institute of Health, Dr. Weber directed funding and studies in the specialty field of women’s health, which includes pelvic floor disorders and prolapse and incontinence. As program director, Dr. Weber helped create the Pelvic Floor Disorders Network, which became one of eight institutions doing data analysis across the U.S..
Dr. Weber also attended the University of Michigan at the School of Public Health, where she received a masters degree in clinical research design and statistical analysis. This refers to clinical research designed to determine how one sets up and performs studies to answer specified questions.
Dr. Weber has published some 100 peer reviewed articles on pelvic floor disorders, according to her testimony.
Plaintiffs’ attorney Adam Slater also preempted defense cross examination by asking Dr. Weber how much money she had made on this case. The answer was about $88,000 in compensation. (The huge expense of litigation is one big reason corporations have pushed so hard for so-called “tort reform,” which now makes many cases cost prohibitive for plaintiffs to pursue. Ms. Budke’s catastrophic injuries in this case makes it worth pursuing).
It was Dr. Weber who authored a bulletin with the focus of protecting women and letting the end user doctors understand mesh repair was not a firmly accepted practice. When, prompted by attorney Slater, she broached the subject of one of the mesh reviewers using the word “experimental,” defense lawyers strenuously objected, as they would some two dozen times during the day.
We know from past testimony that it was Dr. Vincent Lucente who eventually had the word “experimental” removed from the bulletin that doctors would read, a move clearly made to assuage doctors’ fears of liability and also green light insurance coverage.
Alternatives to Experimental Plastic Mesh
Asked to give available therapies for a mild case of incontinence, Dr. Weber detailed various options: first, you could do nothing; then you could use a pessary; do a suture repair (Colporrhaphy) or a biologic or synthetic graft augmented repair; lastly, you could use an experimental mesh kit like the Prolift (which, jurors could not be told, was withdrawn from the market in June 2012).
All of those traditional repairs would be reasonable considering Ms. Budke’s condition, said Dr. Weber. In addition, the Prolift was intended for a stage 3 or 4 prolapse; and Ms. Budke had only a stage one or two. Yes, prolapse might reoccur with a traditional repair, Dr. Weber conceded, but one has to consider how the woman is feeling, consider her overall quality of life. Additionally, more often than not, an older patient with mild prolapse frequently does not see it return, Dr. Weber testified; re-occurrence more often happens among younger patients.
Meanwhile, documents shown to the jury indicated industry-generated data showing a high rate of failure using a traditional repair; hence the reason for pushing the Prolift.
Dr. Weber concluded of the Prolift given Ms. Budke: “In my opinion, it was unnecessary.”
Videotape
Plaintiffs showed videotape ofan Ethicon-created animation of a prolapse procedure as well as an actual videotape of surgery. The defense objected again and the attorneys went to a sidebar with the judge. Then the jury left the courtroom.
The screen shows the roping of the arms in the animation as the cannula leaves the body. Judge Hass almost always granted a defense objection.
With the jury away, Slater argued to the judge: “The video shows roping in real life we are being severely prejudiced and we should have the right to put it in front of the jury. They don’t want the jury to see what they sold.”
After stipulating that Dr. Weber had never seen this procedure in person and had never performed a Prolift surgery, they continued. Dr. Weber was very professorial, wrote Jane Akre, and was comfortable explaining to the 15-person jury who seemed engaged.
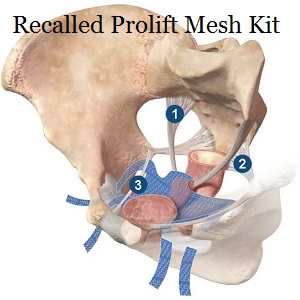
Pelvic organs, FDA
She shows the space where the Prolift mesh is going to be implanted between the front of the vagina (anterior) and the bladder.
About the Prolift implant she said, “The involvement of the hip and the groin would never be involved in a typical repair. In the Prolift, a whole new area is involved that has nothing to do with the prolapse.”
While a company animation showed the mesh arms laying flat, in real life they curled through the cannula, said Dr. Weber, making impossibly small the pore size of the arms in the process.
The Gynemesh PS study
Ethicon had sponsored a study to see what happens when Gynemesh PS is implanted abdominally or through the vagina. It was conducted to encourage doctors to use mesh. In preparation for her expert testimony, Dr. Weber took the time to revisit the study protocol, study collection forms, raw data and information that had been transcribed from a medical chart.
Dr. Weber’s results showed a drastic difference from what Ethicon reported to a scientific committee:
*1 year mesh exposure, same as erosion – Ethicon reported 9.4%
Actual – 15.4% erosion
“That’s a very high rate,” said Dr. Weber.
1 year prolapse recurrence rate 24% Actual – 33.8%
1 year vaginal surgery prolapse recurrence rate (not reported) Actual -27.3%
In addition, doctor consultants working for Ethicon – Dr. Lucente, Dr. Miles Murphy and Dr. Dennis Miller – reported different results at different medical societies, said Dr. Weber.
At the same time the plaintiff’s side showed a document of another mesh being studied with favorable results.
Of the UltraPro (UP) the LIGHTning Human Cadaver Wet Lab participants said:
*Preferred Ultrapro over Gynemesh PS
*Better handling of UP with Prolift instruments
*Feels very soft in place
*No crumpling of the arms
Ethicon knew a Better Mesh Existed
Ethicon knew there was a better mesh was the conclusion. The document is dated September 6, 2006, 17 months before Ms. Budke received her implant.
Inventors balked at High Prolift Fail Rate
Referring to the French inventors of the Prolift TVM group, Dr. Weber said they were not comfortable with having the Prolift used in widespread clinical practice. They felt the erosion rate was too high. “They felt it needed to be under 5% and they were not even close to that.”
What happened to Joan Budke?
Attorney Slater asked Dr. Weber what happened to Joan Budke followed her mesh implant.
Dr. Weber answered that first, she experienced mesh contraction and mesh eroded through the vaginal wall. The vaginal implant naturally picks up bacteria and eventually the infection formed an abscess. The mass was so large it blocked off her ureters, the tubes that run from the kidney to the bladder. With the blockage, she developed acute kidney failure.
In the chronic phase, the white blood cells are fighting the foreign body, there is ongoing cell death and tissue damage. Dr. Weber described a “cascade of events ” – infections, erosions, extrusions, mesh shrinkage, vaginal granulation tissue, sinus formation abscess, fistulas or osteomyelitis (an infection in a bone).
“It’s my opinion she suffered all of these complications,” Dr. Anne Weber.
Ms. Budke lost more than 30 pounds. A foley catheter at one point was implanted to help her urinate. A fistula had developed between her bladder and vagina causing urine to escape that route. Her body began wasting away as she was bed bound. One month before her death in August 2009, doctors considered permanently removing her bladder because it was too destroyed to repair, but Ms. was determined to be too weak to withstand the surgery.
Defective Design
Dr. Weber told the jurors the downhill spiral cannot be reversed unless the mesh is removed, but with a Prolift mesh kit and its many arms, it’s nearly impossible to do so. The mesh was intended to become integrated with the tissue making removal difficult if not possible to remove.
“That’s a design defect.”
At that point the family appeared to be crying, wrote Jane Akre. The two daughters dropped their heads. At least one juror appeared to be tearing up.
“The Prolift was so defectively designed and so unsafe it should never have been on the market,” said Dr. Weber.
Defective design is one key to this product liability case; the other is for plaintiffs to prove that Johnson & Johnson failed to properly warn doctors of the product’s shortcomings.
In this trial, the implanting physician, Dr. Simpson, is also facing medical malpractice.
Related
- $73 Million Mesh Verdict
- Prolift Mesh Recalled
- Transvaginal Mesh Lawsuit
- Transvaginal Mesh Trial for Woman killed by Infection

by Matthews & Associates




