 Amid reports of deaths linked to Essure, a permanent birth control device, an FDA panel has decided to investigate. Law360 reported on June 25, 2015 that an FDA panel will review the safety of Bayer HealthCare AG’s permanent birth control insert Essure.
Amid reports of deaths linked to Essure, a permanent birth control device, an FDA panel has decided to investigate. Law360 reported on June 25, 2015 that an FDA panel will review the safety of Bayer HealthCare AG’s permanent birth control insert Essure.
The agency has received health problem reports not warned of on the product’s label. FDA said at least four women are believed to have died after being implanted with the device. The agency’s Obstetrics and Gynecology Devices Panel plans to meet Sept. 24, 2015 to consider the safety of Essure.
Essure linked to Deaths
The agency has received nearly 5,100 reports of problems related to Essure since its approval in 2002, including four deaths. According to the FDA, one death was linked to an infection after implantation; another to uterine injuries during implantation; another to an embolism during a removal procedure; another to suicide, but noted also that the reports it received could include inaccurate or unverified information, and such reports do not necessarily mean a product is defective.
The FDA’s statement also related reports of five fetal deaths in women who became pregnant despite their Essure implants. The most common problems Essure patients reported to the FDA were abdominal pain, menstrual problems and headaches.
FDA Statement
The agency said in a statement: “Complaints or adverse event reports do not necessarily directly indicate a faulty or defective medical device, and adverse event reports alone cannot be used to establish or compare rates of event occurrence. [We] may receive multiple reports related to the same event, making it difficult to determine actual numbers of events.”
Essure a Permanent Implant
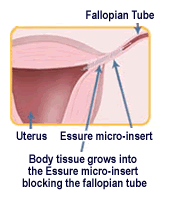
Essure.com says Essure inserts are meant to be permanently implanted into the fallopian tubes to obstruct the ovaries, preventing fertilization. The website calls Essure the only permanent birth control device that doesn’t involve surgery.
Essure Records Manipulated?
Law360 reports that one law firm filed a citizen petition calling for Essure to be pulled from the market, claiming its clinical trial records were manipulated to win Essure’s FDA approval in 2002. One allegation of Essure trial misconduct involves a woman who said she reported severe pain from her Essure implant, yet the trial researchers related her condition as “excellent.” The petition said that another woman in the trial was falsely reported to be 20 years older than her actual age, perhaps to conform with the FDA’s age parameters. Bayer said then that it was still reviewing the petition. The company indicated in a statement that it “is committed to patient safety, and it is our top (priority).”
Related
- Dangerous Medical Devices
- Medical Devices not tested for Safety

by Matthews & Associates




